Fig. 4
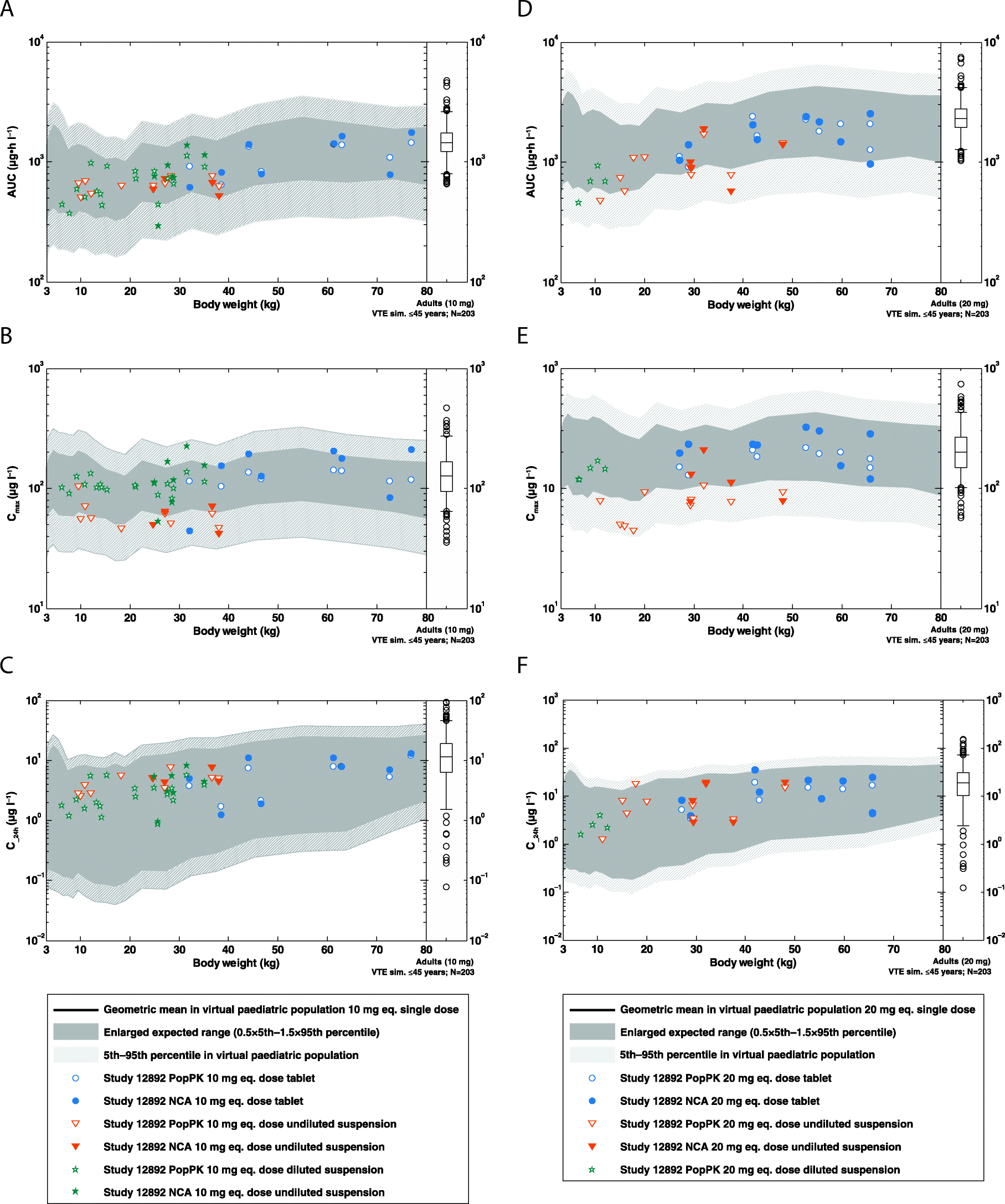
Range plots comparing PK parameters for children aged 0.5–18 years derived from the PopPK analysis or NCA (aged ≥6 years) with the corresponding PBPK model predictions: a AUC0–24, b Cmax and c C_24h for rivaroxaban 10 mg-equivalent doses, and d AUC0–24, e Cmax and f C_24h for rivaroxaban 20 mg-equivalent doses. The solid black line shows the geometric mean of the population prediction and the light grey shaded area denotes the 90% prediction interval of the PBPK model. The dark grey shading denotes the enlarged expected concentration range representing 0.5-times the 5th percentile and 1.5-times the 95th percentile of the PBPK prediction. Data points show PK parameters of individual subjects derived by NCA (closed symbols) or PopPK analysis (open symbols). The corresponding distributions of PK parameters observed via PopPK modelling from an adult reference population (N = 203 adult VTE patients aged 18–45 years) is also shown as box-whisker plot indicating the percentiles 5, 25, 50, 75 and 95 AUC0–24, area under the plasma concentration–time curve from time 0–24 h; C_24h, plasma concentration 24 h after rivaroxaban administration; Cmax, maximal plasma concentration; eq., equivalent; NCA, non-compartmental analysis; PBPK, physiologically based pharmacokinetics; PK, pharmacokinetics; PopPK, population pharmacokinetic; VTE, venous thromboembolism.